Carbon nanotubes9/15/2023  If you want to reproduce the wholeĪrticle in a third-party commercial publication (excluding your thesis/dissertation for which If you are the author of this article, you do not need to request permission to reproduce figuresĪnd diagrams provided correct acknowledgement is given. Provided correct acknowledgement is given. If you are an author contributing to an RSC publication, you do not need to request permission Please go to the Copyright Clearance Center request page. 
To request permission to reproduce material from this article in a commercial publication, 
Provided that the correct acknowledgement is given and it is not used for commercial purposes. This article in other publications, without requesting further permission from the RSC, Nasibulin,Ĭreative Commons Attribution-NonCommercial 3.0 Unported Licence. In vitro toxicity of carbon nanotubes: a systematic review We hope that the present systematic review will shed light on the current knowledge about CNT toxicity, indicate “dark” spots and offer possible directions for the subsequent studies based on the demonstrated here tabulated and statistical data of doses, cell models, toxicity tests, viability, etc. In the case of dispersions, our analysis revealed mean values of dose/incubation time to be 4–5 μg mL −1 h −1, which suggested the material to be a suitable candidate for further studies to get a more in-depth understanding of its properties in biointerfaces and offer CNTs as a promising platform for fundamental studies in targeted drug delivery, chemotherapy, tissue engineering, biosensing fields, etc. We revealed that CNTs located on a substrate had negligible impact, i.e., 90% of studies report good viability and cell behavior similar to control, therefore CNTs could be considered as a prospective conductive substrate for cell cultivation. We also differentiated the impact of various forms of CNTs: on a substrate and in the form of dispersion because in both cases, some studies demonstrated good biocompatibility of CNTs. 
Using several criteria and different scientific databases, we identified and analyzed nearly 200 original publications forming a “golden core” of the field to propose safe doses of the material based on a statistical analysis of retrieved data. To get new momentum for researchers working on the intersection of the biological field and nanomaterials, i.e., CNT materials, we systematically reviewed existing studies with in vitro toxicological data to propose exact doses that yield toxic effects, summarize studied cell types for a more thorough comparison, the impact of incubation time, and applied toxicity tests. Raising the toxicity question again, many research groups conclude low toxicity of the material and its potential safeness at some doses for contact with biological systems. The controversial and disputable data about toxicity doses, proposed hazard effects, and human health concerns significantly restrict CNT applications in biomedical studies, laboratory practices, and industry, creating a barrier for mankind in the way of understanding how exactly the material behaves in contact with living systems. 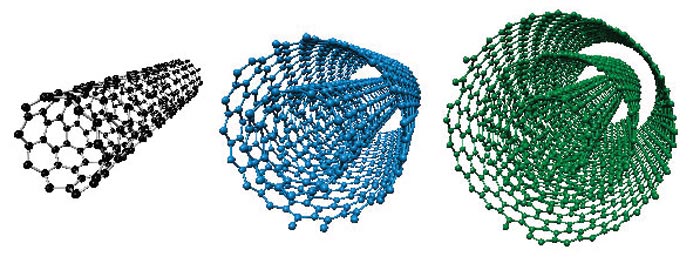
Carbon nanotube (CNT) toxicity-related issues provoke many debates in the scientific community. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
